Regenerative medicine is revolutionizing how we approach musculoskeletal health. This cutting-edge field, backed by leading institutions like the Cleveland Clinic, Mayo Clinic, and the Hospital for Special Surgery, has opened the door to innovative treatments using orthobiologics—therapies derived from your body’s own biological materials to heal and restore orthopedic conditions.
As public interest in regenerative medicine grows, it’s crucial to separate fact from fiction. Informed decisions about your health begin with understanding the science behind these treatments and recognizing the importance of choosing reputable providers.
What You Need to Know
The internet is flooded with information about stem cells, exosomes, amniotic tissue, and umbilical cells. While the FDA plays a critical role in regulating these treatments, some clinics still offer unapproved substances or make misleading claims about their efficacy. Educating yourself is the first step in protecting your health and achieving the best outcomes.
Here’s how to navigate the world of regenerative orthopedics with confidence:
Read more on the establishment of standard procedures in regenerative orthopedics here.
- Check for FDA Compliance
Not all regenerative procedures are the same. The FDA classifies these treatments under two categories:- 361 Procedures: These meet stringent FDA guidelines and involve minimal manipulation of your tissue.
- 351 Procedures: These are more complex, require FDA approval, and are subject to stricter oversight.
Understanding this distinction helps you identify treatments that align with FDA regulations and proven research.
- Scrutinize the Procedure and Provider
Expertise matters. Ask yourself:- Is the provider a licensed physician with specialized training in musculoskeletal medicine?
- Can they explain the research and rationale behind the procedure?
- Are they transparent about outcomes and risks?
Remember, the right provider will prioritize your understanding and safety above all else. Learn why not all regenerative procedures are created equal here.
Stay Informed
Choosing a regenerative orthopedic procedure is a big decision. By understanding the science, recognizing credible providers, and seeking FDA-compliant treatments, you can confidently explore options that support your health and well-being. If something doesn’t feel right or a provider can’t answer your questions, don’t hesitate to walk away and look for a trusted expert.
False Claims in Regenerative Medicine
The surge of unapproved regenerative procedures advertised online is a growing concern. Many of these treatments lack the robust clinical evidence required for FDA approval, making it critical for patients to approach such claims with caution.
The Truth About “Stem Cell” Products
One widespread issue is the misrepresentation of amniotic tissue products. While marketed as containing live stem cells, research, such as the study by Fortier et al. (2021), linked here, has shown that most of these products do not contain live cells. During processing—whether through freezing or drying—the cells are rendered nonviable. Despite this, some manufacturers continue to promote these products to uninformed providers, perpetuating misinformation.
FDA’s Role in Protecting Patients
The FDA has taken a strong stance on the marketing and use of unproven “stem cell” therapies. Their regulatory oversight hinges on a key question: does the injected substance maintain the natural integrity of the harvested tissue, or does it qualify as a “drug”?
To gain FDA approval, regenerative procedures must meet four specific criteria that ensure the biologic product's safety, effectiveness, and integrity. These criteria serve as the benchmark for distinguishing credible, compliant treatments from those that are unproven or unsafe.
Be an Informed Consumer
When evaluating regenerative procedures, it’s vital to scrutinize the claims made by clinics and manufacturers. Ask for evidence of FDA approval, and don’t hesitate to consult studies or seek second opinions. By staying informed, you can avoid falling victim to false claims and prioritize treatments backed by science.

FDA Oversight on Exosome Products and Other Biologics
Exosome products have garnered significant attention in regenerative medicine, but it’s crucial to understand their regulatory status. The FDA requires exosome products intended for the treatment of diseases or conditions in humans to gain FDA approval. As of now, there are no FDA-approved exosome products for such purposes.
FDA Consumer Alerts on Unapproved Procedures
The FDA has flagged several unapproved procedures involving biologic substances. These include:
- Stromal vascular fraction (fat-derived cells)
- Umbilical cord fluid/tissue
- Amniotic fluid/tissue
- Exosomes
Patients should exercise caution when encountering clinics offering these unapproved therapies, as their safety and efficacy remain unproven.
The Role of the FDA in Regulating Biologic Substances
The FDA’s authority to regulate human biologic materials has been upheld in significant legal cases. One notable example involved stromal vascular fraction (SVF), a procedure that isolates fat-derived cells for use in treatments. A California court initially ruled against the FDA’s right to regulate this process, claiming patient cells could not be classified as a drug. However, this decision was unanimously overturned on appeal. The higher court reinforced the FDA’s authority, affirming its critical role in overseeing the use of human biologics to protect patient safety.
What This Means for Patients
Understanding the regulatory landscape is essential when considering regenerative treatments. The FDA plays a vital role in ensuring that therapies are safe, effective, and backed by rigorous research. If you’re exploring options for regenerative medicine, look for treatments that meet FDA standards and avoid clinics promoting unapproved procedures.
What Procedures Are Approved?
At NTXMSK, we adhere strictly to FDA-compliant treatment protocols, ensuring patient safety and effectiveness. Two widely accepted and approved procedures under FDA guidelines are Platelet-Rich Plasma (PRP) Injections and Bone Marrow Aspirate Concentrate (BMAC). These treatments are recognized for their ability to promote healing and regeneration while maintaining compliance with minimal tissue manipulation standards.
- Autologous (sourced from patient’s own body)
- Minimal manipulation
- Homologous use (use is consistent with natural purpose of cell)
PRP and BMAC: Why They Work
- PRP (Platelet-Rich Plasma): A concentration of platelets and growth factors derived from your own blood to accelerate healing in tissues.
- BMAC (Bone Marrow Aspirate Concentrate): Harvested from bone marrow, this treatment preserves viable stem cells, growth proteins, and nutrients by adhering to FDA’s minimal manipulation guidelines.
Minimal processing ensures that the natural integrity and functionality of the tissue are maintained, making these procedures both compliant and effective.
Evidence Supporting BMAC Over Amniotic Products
A 2019 study by Panero et al. (read here) highlights critical findings regarding mesenchymal stem cells (MSCs) in regenerative therapies:
- MSCs were not found in commercial amniotic fluid products (AFPs) or unprocessed amniotic fluid.
- MSCs were successfully cultured from bone marrow aspirates, confirming their viability in treatments like BMAC.
This underscores the importance of using scientifically validated sources, such as bone marrow aspirates, over commercial amniotic products often marketed with misleading claims.
FDA Approval and Clinical Evidence
For any regenerative procedure to gain FDA approval, it must demonstrate safety and efficacy through rigorous clinical trials. Both PRP and BMAC have undergone such trials and consistently shown positive outcomes. The Panero study also compared the results of PRP and Wharton’s Jelly (an amniotic tissue product), further emphasizing the superior performance and compliance of PRP and BMAC.
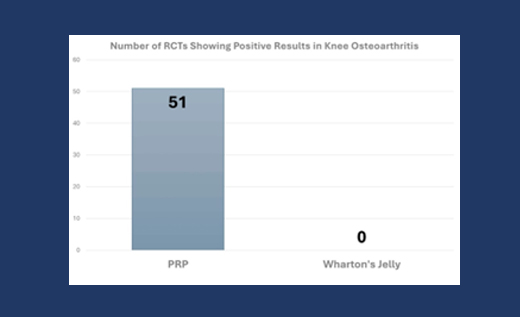
Panero et al.
Proven Effectiveness of PRP and BMAC
Published randomized clinical trials confirm that Platelet-Rich Plasma (PRP) and Bone Marrow Aspirate Concentrate (BMAC) yield favorable outcomes for patients. These studies demonstrate not only the safety but also the effectiveness of these FDA-compliant regenerative procedures in promoting healing and improving musculoskeletal health.
Why PRP and BMAC Are Trusted Options
The positive results from these clinical trials provide a strong foundation for the use of PRP and BMAC in regenerative medicine. Their ability to harness your body’s natural healing processes—through the concentration of platelets, growth factors, and viable stem cells—has made them reliable and scientifically validated options for promoting healing and improving musculoskeletal health in a wide range of orthopedic conditions.
Explore Your Treatment Options
Taking the first step towards improving your musculoskeletal health starts with understanding your treatment options. Learn more about how PRP and BMAC can help you achieve lasting results through safe and effective regenerative therapies.
Click here to access a curated list of research studies and explore the science behind these innovative treatments.
